|
Integer spin particles, bosons, are not subject to the Pauli exclusion principle: any number of identical bosons can occupy the same quantum state, as with, for instance, photons produced by a laser and Bose–Einstein condensate. A more precise way to describe the Pauli. Only fermions (examples are protons, neutrons and electrons) are bound by the Pauli exclusion principle, while bosons (an example is a photon - light beam) are not. The Pauli exclusion principle says that the four Quantum numbers of two electrons of an electron can never be the. The Pauli exclusion principle refers to the fact that certain particles cannot be at the same place at the same time, with the same energy. This means that the wave function changes its sign if the space and spin co-ordinates of any two particles are interchanged. Definition of the Pauli Exclusion Principle. Consistent with Pauli’s exclusion principle, no two rows of the table have the exact same set of quantum numbers. A more rigorous statement is that the total wave function for two identical fermions is anti-symmetric with respect to exchange of the particles. The uncertainty principle is a trade-off between two complementary variables, such as position and speed. Right: As the rollercoaster car descends the hill, we can measure its speed over time but would be less certain about its position. While the earlier descriptions of the hydrogen molecular ion and hydrogen molecule produced many important observations about chemical bonds, they are highly misleading in one aspect. But the snapshot alone would not give us enough information about its speed. This principle was formulated by Austrian physicist Wolfgang Pauli in 1925. Before proceeding to a description of chemical bonds, one important point must first be made. 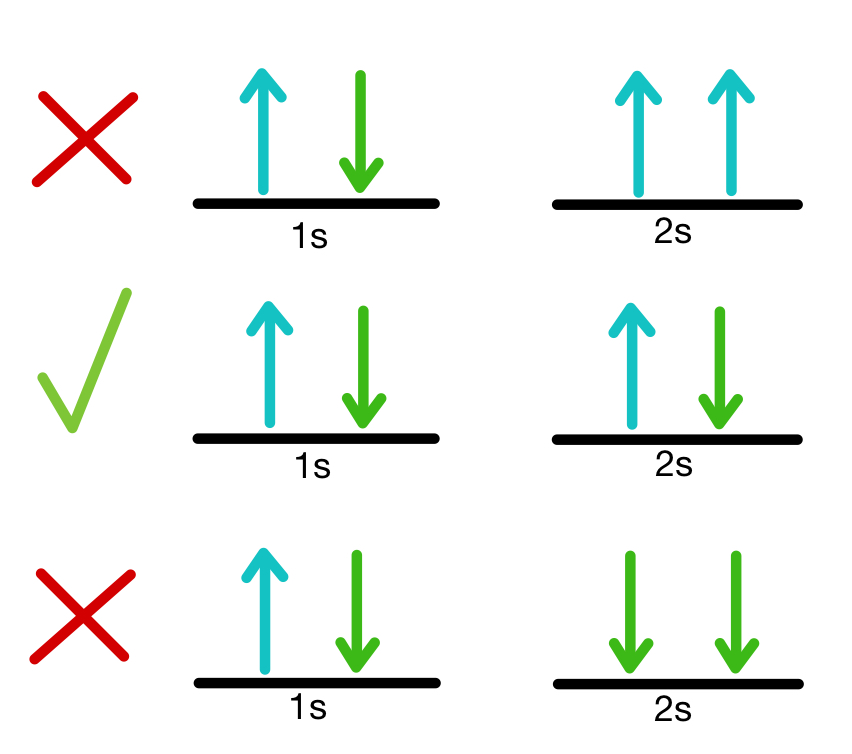
For two electrons residing in the same orbital, n, ℓ, and mℓ are the same, so ms must be different and the electrons have opposite spins. The Pauli Exclusion Principle states that no two identical fermions may occupy the same quantum state in an atom simultaneously. 
In the case of electrons, it can be stated as follows, It is impossible for two electrons of a poly-electron atom to have the same values of the four quantum numbers. Pauli Exclusion Principle definition: One orbital can contain only two electrons, and they must have opposite spins. 
Paulis exclusion principle and Hunds rule, electronic configuration of. Pauli exclusion principle The Pauli exclusion principle is the quantum mechanical principle that two identical fermions cannot occupy the same quantum state simultaneously. The force between two parallel currents carrying conductors-definition of ampere.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
